World Cancer Research Day
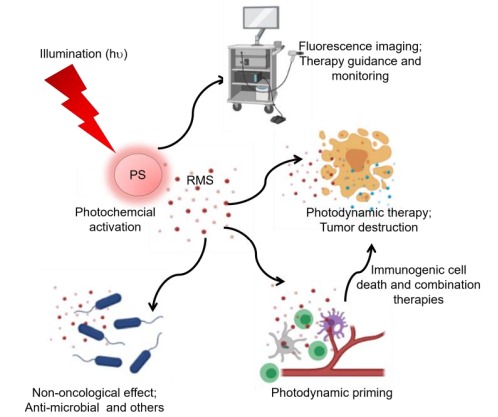
Light activation of materials initiates thermal, photochemical, and/or radiative processes that can be harnessed for response-based therapeutic design, diagnostics, and therapy monitoring. Photodynamic therapy (PDT) leverages photochemistry-based processes and involves the exposure of light activatable molecules to appropriate wavelengths of light resulting in the generation of reactive molecular species which are responsible for targeted cell/ tissue death. The typical molecules used for PDT also have a finite fluorescence quantum yield, so the same entity provides an imaging handle, thus, making photodynamic activation a natural theranostic technology. Furthermore, the ability to use light as a reagent to trigger several downstream events such as drug release allows for the fabrication of intelligent multiagent macromolecules/nanomaterials, antibody conjugates, and other photoresponsive formulations that attack multiple pathways with spatio-temporal control. The photodynamic process is by necessity a multidisciplinary platform that integrates diverse expertise across chemistry, biology, engineering, and clinical science that the current PDT community possesses.
Photodynamic therapy has been approved as a local therapy by regulatory authorities worldwide for over 20 indications since 1995. Additionally, there are encouraging results from a variety of clinical studies based on exquisite mechanistic understanding of the photodynamic activation process. Such mechanistic advances enable deliberately designed combination treatments and inspire the creation of new combinatorial controllable entities, including repurposing of drugs.
Despite significant scientific progress, regulatory approvals for almost 3 decades and being a minimally invasive and effective modality (sometimes when all other therapies have failed), PDT has not yet had the impact it deserves. Approvals aside, the adoption of PDT into medical practice has been slow with a few exceptions. The reason for this is not clear and is probably multifaceted. This may be due to real or imagined complexity of needing a drug and a device, competition with established and financially more lucrative options that require multiple applications (thus providing high profits), lack of efforts at educating healthcare personnel about the technology, and in the case of cancer and perhaps infections, the perceived limitation of it being a localized therapy.
Photodynamic activation can also have a priming effect, extending PDT beyond a purely localized modality and positioning it as a strategy with the potential for systemic disease management. The priming process, whether it be photodynamic (PDP) or a biochemical (such as pretreatment with Vitamin D), the targets are primed for a mechanism-based second treatment within a combinatorial mix. An exciting effort these days is the use of PDT/PDP for increase in stromal and vascular permeability thereby improving drug delivery or enabling immune cells to enter immunologically “cold” tumors making them candidates for immune check point inhibitors. This concept is in clinical trials in Pancreatic Cancer treatment. Such immunological effects have been attributed to the observed abscopal effects associated with PDT and can be attributed to depletion of immune suppressive regulatory T-cells and reprogramming of macrophages to an anti-tumorigenic phenotype.
Recent efforts toward the implementation of PDT have been bold and innovative and develop it beyond local disease control. The photodynamic activation process is versatile and potentially rich with possibilities and if exploited cleverly could lead to high impact treatments and technologies. Its intrinsic theranostic nature, combining therapeutic efficacy with imaging capability, positions PDT as a powerful tool for patient-tailored treatment strategies. Coupled with progress in optical technologies and the emergence of affordable light sources and detection systems, PDT is increasingly accessible. This accessibility makes it particularly well-suited for adoption in low-resource settings with a high cancer burden, especially when integrated with portable, smartphone-based platforms.
As our understanding of the mechanisms underlying photodynamic activation continues to expand, PDT is increasingly recognized as an enhancer of both established and emerging therapies, with strong potential in precision medicine. Rather than competing with conventional modalities such as radiation or chemotherapy, PDT complements them, serving to augment their effectiveness.
Relevant references
- Y. Hanada, S. P. Pereira, B. Pogue, E. V. Maytin, T. Hasan, B. Linn, T. MangelsDick, K. K. Wang, EUS-guided verteporfin photodynamic therapy for pancreatic cancer. Gastrointestinal Endoscopy 94,
179-186 (2021). - G. Obaid, J. P. Celli, M. Broekgaarden, A.-L. Bulin, P. Uusimaa, B. Pogue, T. Hasan, H.-C. Huang, Engineering photodynamics for treatment, priming and imaging. Nature Reviews Bioengineering 2, 752-769 (2024).
- Y. Maruoka, H. Wakiyama, P. L. Choyke, H. Kobayashi, Near infrared photoimmunotherapy for cancers: A translational perspective. eBioMedicine 70, (2021).
- B. Q. Spring, A. O. Abu-Yousif, A. Palanisami, I. Rizvi, X. Zheng, Z. Mai, S. Anbil, R. B. Sears, L. B. Mensah, R. Goldschmidt, S. S. Erdem, E. Oliva, T. Hasan, Selective treatment and monitoring of disseminated cancer micrometastases in vivo using dual-function, activatable immunoconjugates. 111, E933-E942 (2014).
- S. A. Siddiqui, S. Siddiqui, M. A. B. Hussain, S. Khan, H. Liu, K. Akhtar, S. A. Hasan, I. Ahmed,
S. Mallidi, A. P. Khan, F. Cuckov, C. Hopper, S. Bown, J. P. Celli, T. Hasan, Clinical evaluation of a
mobile, low-cost system for fluorescence guided photodynamic therapy of early oral cancer in India.
Photodiagnosis and Photodynamic Therapy 38, 102843 (2022). - B. Q. Spring, R. Bryan Sears, L. Z. Zheng, Z. Mai, R. Watanabe, M. E. Sherwood, D. A.
Schoenfeld, B. W. Pogue, S. P. Pereira, E. Villa, T. Hasan, A photoactivable multi-inhibitor
nanoliposome for tumour control and simultaneous inhibition of treatment escape pathways. Nature
Nanotechnology 11, 378-387 (2016). - S. Mallidi, K. Watanabe, D. Timerman, D. Schoenfeld, T. Hasan, Prediction of Tumor
Recurrence and Therapy Monitoring Using Ultrasound-Guided Photoacoustic Imaging. Theranostics 5,
289-301 (2015). - M. Overchuk, K. M. Harmatys, S. Sindhwani, M. A. Rajora, A. Koebel, D. M. Charron, A. M.
Syed, J. Chen, M. G. Pomper, B. C. Wilson, W. C. W. Chan, G. Zheng, Subtherapeutic Photodynamic
Treatment Facilitates Tumor Nanomedicine Delivery and Overcomes Desmoplasia. Nano Letters 21,
344-352 (2021).