Water, both common and necessary for life on Earth, behaves very strangely in comparison with other flowing substances. How water’s properties such as density, specific heat, viscosity and compressibility respond to changes in pressure and temperature is completely opposite to other liquids that we know. Consequently, water is often called “anomalous”. If water would have behaved as a “normal liquid” we would not exist, since marine life could not have developed.
All matter shrinks when cooled, resulting in an increase in density. Therefore, one would expect water to have a high density at its freezing point. However, as we all know, ice cubes float, so everything is upside down when we look at a glass of ice water (Fig. 1). Strangely enough for the liquid state, water is the densest at 4 degrees C, and therefore it stays on the bottom whether it’s in a glass or in an ocean. If you chill water below 4 degrees, it starts to expand again. If you continue to cool pure water (where the rate of crystallization is low) to below 0 degrees, it continues to expand – the expansion even speeds up when it gets colder.

If water were an ordinary liquid, ice would sink to the bottom and the water would be densest at 0°C. The bottoms of oceans and lakes would freeze in winter and take a long time to thaw in summer, which would have prevented life from evolving. The maximum density also provides circulation of nutrients in the ocean and lakes, since if the water at the bottom becomes cooler than 4°C, it floats up towards the surface. These properties have enabled marine life, and eventually mammals including humans, to evolve on Earth.
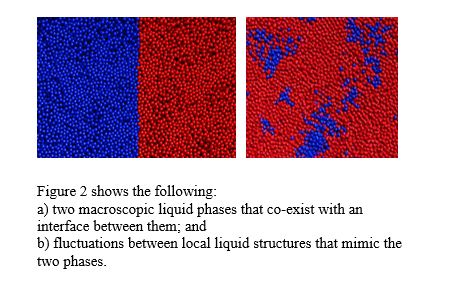
We now understand the origin of the strangeness. Water is unique in that it can exist in two macroscopic liquid phases with different bonding properties of water molecules at low temperatures and high pressures (Fig. 2a)1, 2. As the temperature increases and the pressure decreases, a state is reached where the distinction between the two liquid phases vanishes and only one phase remains3. This is a highly unstable point, causing fluctuations in a large temperature and pressure region, extending all the way up to ambient conditions4. Water fluctuates between the two liquid states and mixtures of the two, as if it cannot make up its mind (Fig. 2b). These fluctuations give water its unusual properties. The state beyond the critical point is called supercritical, and this is the state of ambient water.
It is intriguing that water is the only supercritical liquid at ambient conditions where life exists and we also know there is no life without water. Is this a pure coincidence or is there some essential knowledge for us to gain in the future?
References
- P. H. Poole, F. Sciortino, U. Essmann and H. E. Stanley, Nature 360 (6402), 324-328 (1992).
- K. H. Kim, K. Amann-Winkel, N. Giovambattista, A. Späh, F. Perakis, H. Pathak, M. L. Parada, C. Yang, D. Mariedahl, T. Eklund, T. J. Lane, S. You, S. Jeong, M. Weston, J. H. Lee, I. Eom, M. Kim, J. Park, S. H. Chun, P. H. Poole and A. Nilsson, Science 370, 978-982 (2020).
- P. G. Debenedetti, F. Sciortino and G. H. Zerze, Science 369, 289-292 (2020).
- K. H. Kim, A. Späh, H. Pathak, F. Perakis, D. Mariedahl, K. Amann-Winkel, J. A. Sellberg, J. H. Lee, S. Kim, J. Park, K. H. Nam, T. Katayama and A. Nilsson, Science 358, 1589–1593 (2017).
Professor of Chemical Physics, Stockholm University, Sweden.
Recall Professor Emeritus in Photon Science Stanford University, USA.
Anders Nilsson received a PhD in physics at Uppsala University, Sweden (1989) in the laboratory created by the Nobel Laureate Kai Siegbahn. Before returning to Sweden in 2014 he was 15 years as a professor in Photon Science at Stanford University.
He received the Plyler Prize at the American Physical Society, Lindbomska Award at the Swedish Royal Academy of Science, the Royal Oscar Award at Uppsala University, the Shirley Award in Berkeley, the Humboldt Award for senior scientist in Germany and was awarded Honorary Doctorate at Denmark’s Technical University.
The research on water was selected in 2004 by Science Magazine as one of the top ten breakthrough of the year, illustrated in 2014 as cover of Nature, selected as one of the 100 most essential discoveries in 2017 by Discovery Magazine and illustrated with cover in New Scientist in 2018. His research interests include x-ray spectroscopy and scattering, chemical bonding and reactions on surfaces, ultrafast science, heterogeneous catalysis, electrocatalysis in fuel cells, photocatalysis for converting sunlight to fuels, structure, dynamics and physical properties of water and aqueous solutions.


